AioCare Patient is a personal application for patients who suffer from asthma, COPD, cystic fibrosis (CF), idiopathic pulmonary fibrosis (IPF) and all, who want to constantly monitor their lungs
AioCare Patient



What is it about?
AioCare Patient is a personal application for patients who suffer from asthma, COPD, cystic fibrosis (CF), idiopathic pulmonary fibrosis (IPF) and all, who want to constantly monitor their lungs. It is intended to use with portable spirometer AioCare.

App Screenshots





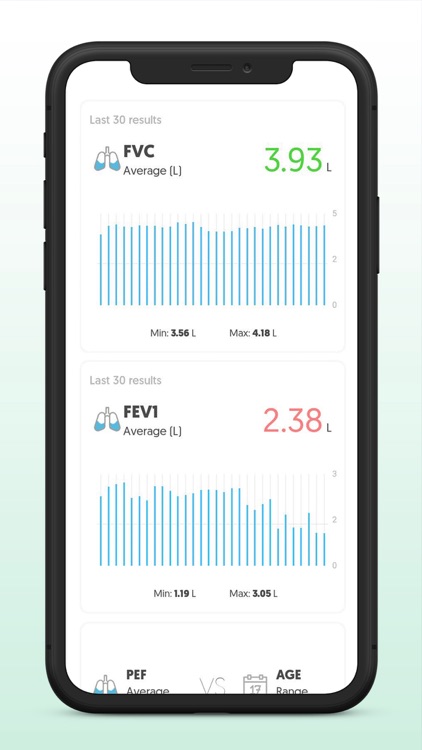
App Store Description
AioCare Patient is a personal application for patients who suffer from asthma, COPD, cystic fibrosis (CF), idiopathic pulmonary fibrosis (IPF) and all, who want to constantly monitor their lungs. It is intended to use with portable spirometer AioCare.
Use the digital diary to record symptoms and accurately measure lung function with the portable AioCare spirometer and share it in real-life with your doctor.
Ultimate features of AioCare Patient app:
- Full-loop spirometry (FEV1, FVC, FEV1/FVC)
- Peak Expiratory Flow testing (PEF)
- Heart rate module (medical grade)
- ATS/ERS Standards of Spirometry
- Automatic quality check (AI based algorithms)
- Print, download or send results as PDF or Excel
- Convenient contact with your Doctor via secure chat
- Spirometry test reminders (app notifications)
- Secure health data storage (on-line & off-line)
- E-mail support within 48 hours
Coming soon
- Oximeter
- Asthma Action Plan
- Self-management plan in the event of an exacerbation - Disease Action Plan planned by your doctor
- Slow vital capacity (SVC maneuver)
AioCare enables to optional and safe synchronize your measurements results with the Apple Health App thus you can keep all your health information in one place and monitor your health better!
You decide what do want to synchronize:
- Height
- Weight
- Heart Rate
- Forced Expiratory Volume, 1 sec
- Forced Vital Capacity
- Peak Expiratory Flow Rate
Version for Patients.
Note: The application works with a class IIa medical device that meets the medical device (MDD) standards, compliant with ISO 26782: 2009 and ISO 23747: 2015 standards. The device is certified and CE marked by the TUV Nord Accredited Certification Body for medical devices in the European Economic Area.
Please use carefully and in accordance with the purpose and territory (EEA/EU) and in accordance with the instructions for use.
AppAdvice does not own this application and only provides images and links contained in the iTunes Search API, to help our users find the best apps to download. If you are the developer of this app and would like your information removed, please send a request to takedown@appadvice.com and your information will be removed.